|
The same kind of backbonding occurs with phosphine complexes, which have empty π orbitals, as shown at the right. The C-O infrared stretching frequency is diagnostic of the strength of the bond and can be used to estimate the degree to which electrons are transferred from the metal d-orbital to the CO π-antibonding orbital. This interaction strengthens the metal-carbon bond but weakens the carbon-oxygen bond. In metal carbonyl complexes such as Ni(CO) 4 and Mo(CO) 6, there is sideways overlap between filled metal d-orbitals and the empty π-antibonding orbitals (the LUMO) of the CO molecule, as shown in the figure below. Pπ-dπ bonding is also important in transition metal complexes. For example, phosphines (R 3P:) are good σ donors in complexes with transition metals, as shown below. Transition metal d-orbitals can also form σ bonds, typically with s-p hybrid orbitals of appropriate symmetry on ligands. Compounds with metal-metal δ bonds occur in the middle of the transition series. δ bonds are generally quite weak compared to σ and π bonds. For this reason, compounds containing C=C double bonds are very common, but those with Si=Si bonds are rare. 
π-bonded compounds of heavier elements are rare because the larger cores of the atoms prevent good π-overlap. 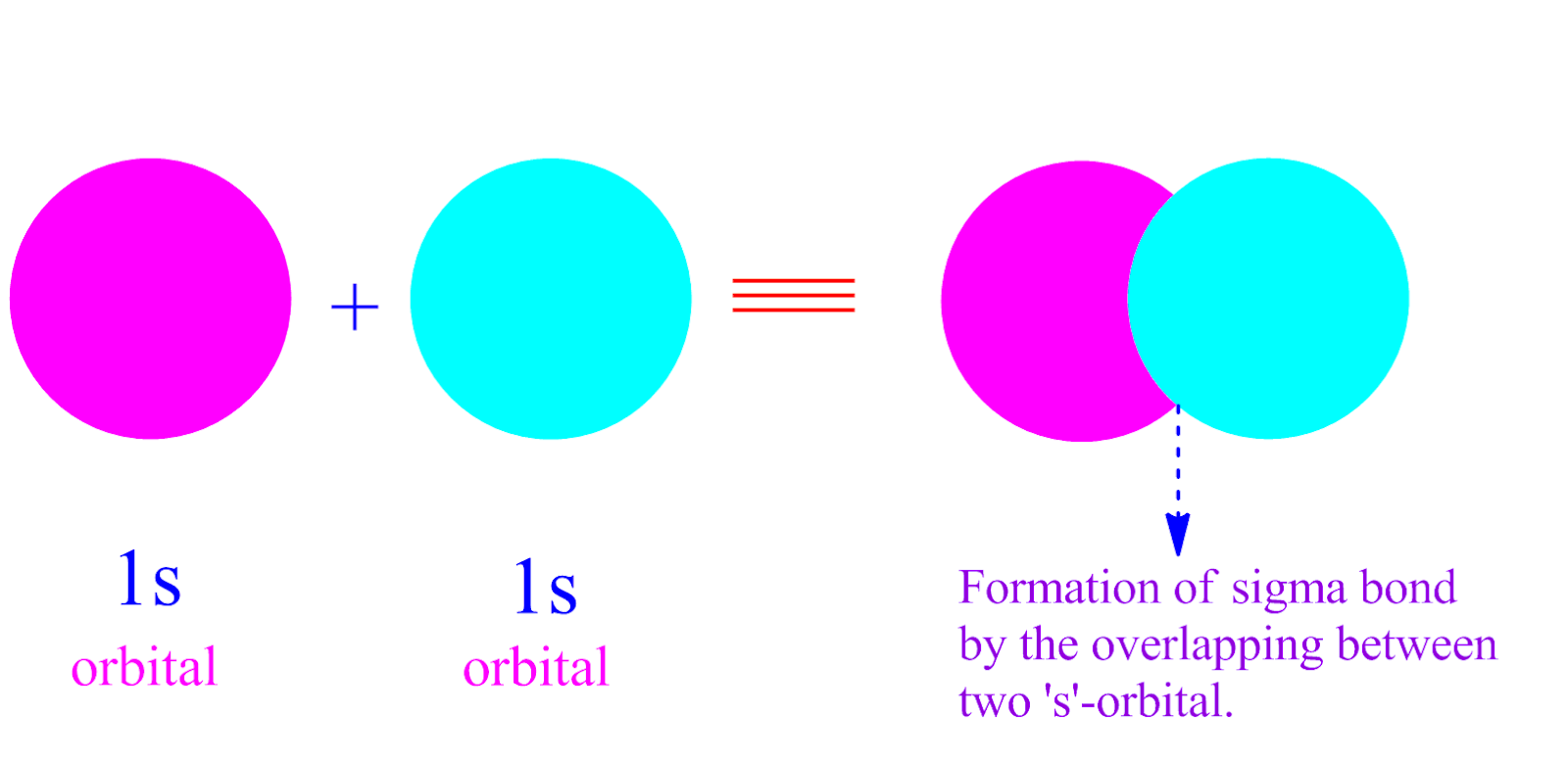
Because pπ-pπ bonding involves sideways overlap of p-orbitals, it is most commonly observed with second-row elements (C, N, O). In each case, we can make bonding or antibonding combinations, depending on the signs of the AO wavefunctions. Some possible σ (top row), π (bottom row), and δ bonding combinations (right) of s, p, and d orbitals are sketched below. \): The octachlorodirhenate(III) anion, 2−, which has a quadruple Re-Re bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
